 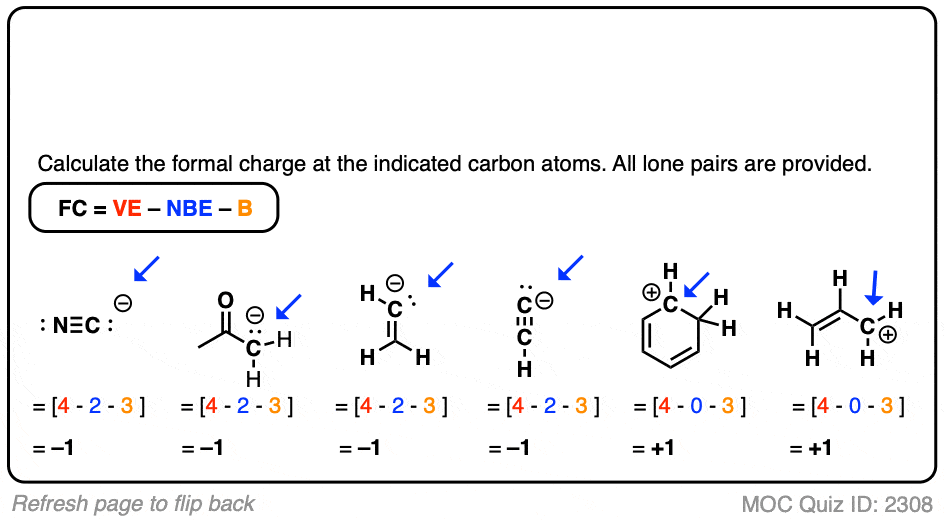 
Any formal charge, any negative, any negative formal charge on individual atom, individual atom, ideally, ideally on most electronegative ones, or most electronegative one. Talking about the charge of the entire ion. So any formal charge, so once again, we're not On the most electronegative of the atoms. And then the electronegativity is useful because we also want to see if there's any negative formal charge on an individual atom that ideally, that would be We're talking about individualĪtoms' formal charges, close to zero as possible. We're not talking about theĬharge of the entire ion. Individual, individual atoms have formal charge as close to zero as possible. The resonance structures where individual atoms have formal charges as close to zero as possible. Structures contributes most to the resonance hybrid? One, we want to figure out We want to optimize for when we're thinking about which of these resonance Structures contributes most to the resonance hybrid of thiocyanate? So pause this video and see Pauling scale electronegativity, so maybe that is going to be useful for thinking about basing on the, based on the formal charges, which of the three 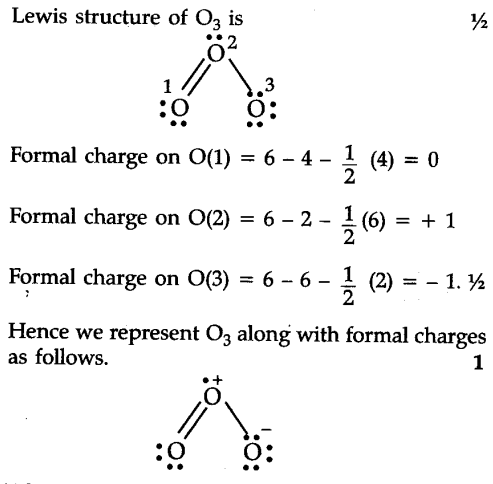
They've given us the various elements in these resonance structures, and they've told us their Structures contributes most to the resonance hybrid of thiocyanate? And they have given us Based on formal charges, which of the three We're told that three possible resonance structures for the thiocyanate ion are shown below. If a formal charge is unavoidable than resonance structures with negative formal charges on more electronegative atoms like oxygen or nitrogen is more stable and helps the resonance structures contribution to the hybrid. Charge separation being formal charges on atoms where they would normally like to be neutral. A resonance structure having less charge separation helps it contribute more because charge separation creates instability. Having filled octets helps a resonance structure contribute more to the resonance hybrid because having filled octets is more stable than not having them filled. Now the resonance structures have different stabilities and contribute unequally to the resonance hybrid which is what this question is asking about. The combination of all the resonance structures is what the molecule looks like and is called the resonance hybrid. Now for actually predicting what the molecules looks like which one of the resonance structures is the correct one? Well they all are to an extent. They have the same atoms, the same connections, the same overall charge, but where the valance electrons are different between the resonance structures. However some molecules have more than 1 valid Lewis structure and we call those resonance structures. The geometry of Molecules is a platform where we try to educate you about various chemistry concepts and find the answers you have been looking for.So, Lewis structures are just how we can model the structure of molecules by placing the valance electrons of the atoms. And it might seem challenging, but it becomes the most fun and easy subject to learn if learned the correct way. Sounds easy, right? Go ahead and try our formal charge calculator.ĭo you know that a perfect diamond is made up of a single Carbon molecule? Chemistry is one of the most exciting subjects. And to make it easy for you to do the calculations we have developed a Formal charge calculator for you, where you have to just enter the number of valence electrons, non-bonding electrons, and bonding electrons to find out the formal charge. Similarly, you can use this formula for other molecules and find out formal charges for individual atoms in the molecule. And using the formula stated above, we found out that the total charges on both Sulphur and Fluorine atoms are zero. Here in SF4 as all the Fluorine atoms are arranged symmetrically, they will have the same charges. of valence electrons Non-bonding electrons Bonding electrons Total charge Sulphur (S) Now that we know the formula let us look at the example of how to find out formal charges for individual atoms in a polyatomic molecule Let us calculate the formal charges of SF4 Atom Total no. One can calculate the formal charges for any given atom with the help of the following formula:į.C = Valence electrons – Nonbonding electrons- Bonding electrons/2 These charges help in knowing if the given structure of the molecule is stable or not. 
Formal charge is the individual electric charges on the atoms in a given polyatomic molecule. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
